
Report ID: UCMIR35G2187
SkyQuest Technology's U.s. in-vitro diagnostics market size, share and forecast Report is based on the analysis of market data and Industry trends impacting the global U.S. In-vitro Diagnostics Market and the revenue of top companies operating in it. Market Size Data and Statistics are based on the comprehensive research by our Team of Analysts and Industry experts.
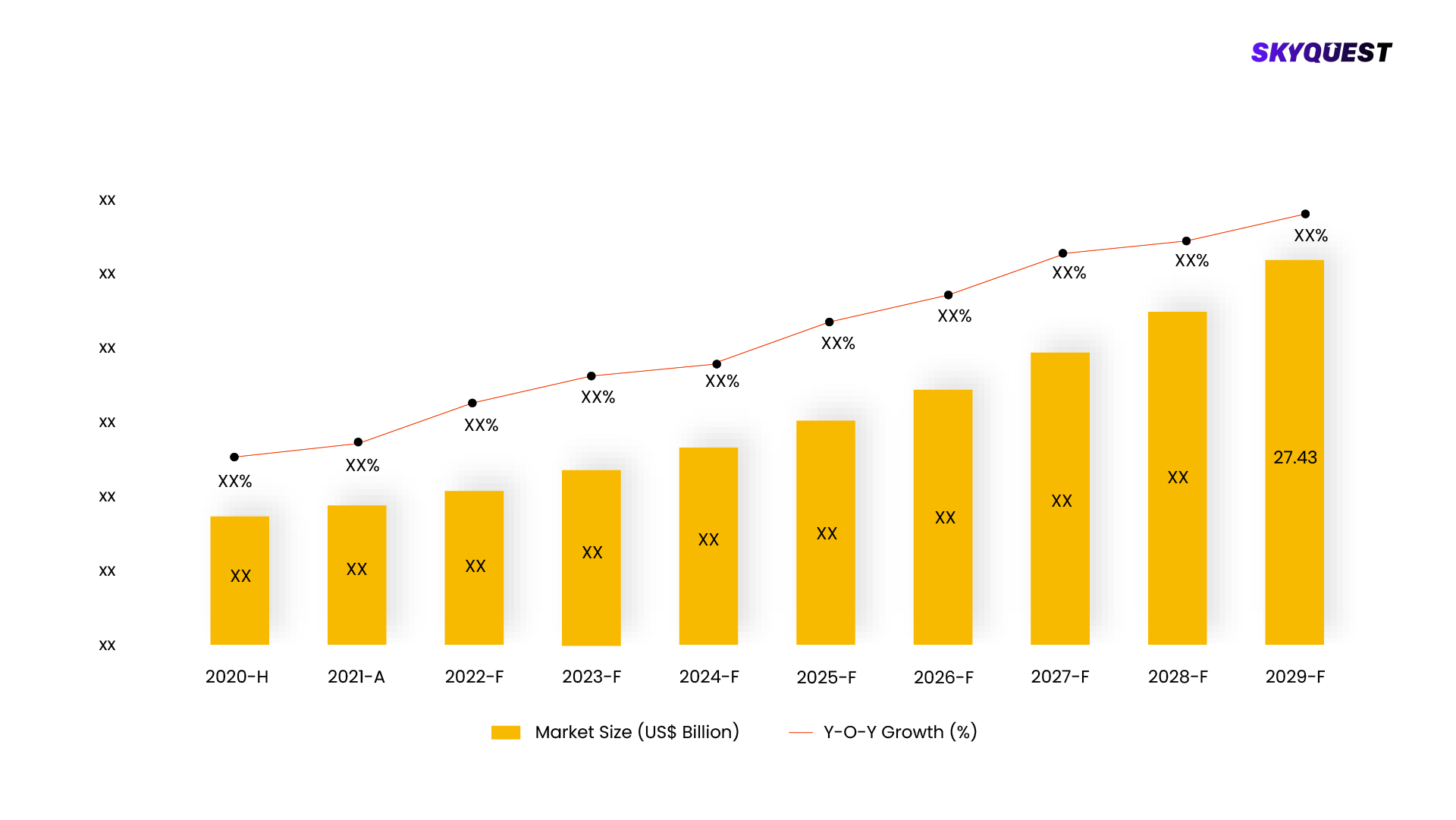
In 2022, the United States' in-vitro diagnostics sector recorded a market size of USD 32.94 billion. Forecasts indicate a significant expansion, with expectations that the market will increase from USD 33.78 billion in 2023 to USD 56.81 billion by 2030, reflecting a robust compound annual growth rate (CAGR) of 7.7% throughout the forecast period. This growth is attributed to the substantial presence of prominent industry players, a rising demand for quick diagnostic testing and point-of-care devices, an increasing incidence of chronic illnesses, and heightened awareness concerning early disease detection.
This report is being written to illustrate the market opportunity by region and by segments, indicating opportunity areas for the vendors to tap upon. To estimate the opportunity, it was very important to understand the current market scenario and the way it will grow in future.
Production and consumption patterns are being carefully compared to forecast the market. Other factors considered to forecast the market are the growth of the adjacent market, revenue growth of the key market vendors, scenario-based analysis, and market segment growth.
The market size was determined by estimating the market through a top-down and bottom-up approach, which was further validated with industry interviews. Considering the nature of the market we derived the Health Care Technology by segment aggregation, the contribution of the Health Care Technology in Health Care Equipment & Services and vendor share.
To determine the growth of the market factors such as drivers, trends, restraints, and opportunities were identified, and the impact of these factors was analyzed to determine the market growth. To understand the market growth in detail, we have analyzed the year-on-year growth of the market. Also, historic growth rates were compared to determine growth patterns.
REQUEST FOR SAMPLE
Want to customize this report? This report can be personalized according to your needs. Our analysts and industry experts will work directly with you to understand your requirements and provide you with customized data in a short amount of time. We offer $1000 worth of FREE customization at the time of purchase.
Feedback From Our Clients

Report ID: UCMIR35G2187
sales@skyquestt.com
USA +1 351-333-4748