
Report ID: SQMIG35D2319

Report ID: SQMIG35D2319
[email protected]
USA +1 351-333-4748

Report ID:
SQMIG35D2319 |
Region:
Global |
Published Date: December, 2025
Pages:
198
|Tables:
187
|Figures:
77



Global Molecular Diagnostics Market size was valued at USD 19.1 Billion in 2024 and is poised to grow from USD 20.49 Billion in 2025 to USD 36.01 Billion by 2033, growing at a CAGR of 7.3% during the forecast period (2026–2033).
The Molecular Diagnostics Market is growing strong, fueled by a boost in demand for precision medicine and early disease identification. Among the most important drivers is the rising incidence of infectious diseases and cancers worldwide, which calls for swift and accurate diagnostic technologies. Technological advances—real-time PCR, next-generation sequencing (NGS), and microarray methods—are making for speedier, more accurate identification of pathogens and genetic mutations. Also contributing to the expansion of the market is the increased use of companion diagnostics to direct targeted treatments in oncology and other chronic diseases.
The increasing use of molecular diagnostics in daily clinical practice, particularly in developed markets, is also augmenting the growth of the market.Another force driving the market is the increasing trend of point-of-care (POC) testing and at-home diagnostics, fueled by the COVID-19 pandemic. This trend has led to considerable investment in mobile and user-friendly diagnostic platforms. In addition, rising healthcare expenditure, auspicious government policies, and improving awareness of early disease detection are driving market growth. Expansion of personalized medicine and emphasis on genomics within global healthcare systems are poised to drive long-term demand for molecular diagnostics, particularly in oncology, infectious diseases, and genetic disorder testing.
From the restraint side, there are challenges in the form of high expenses related to molecular diagnostic tests and devices, which may restrain adoption in cost-conscious and emerging markets. The complicated regulatory environment and requirement for experienced manpower to run advanced testing platforms are other restraints that can prevent extensive implementation. Reimbursement policies in various countries are another restrain that acts as a barrier to market entry. In addition, issues surrounding data privacy and the interpretation of highly complex genetic data could limit wider use in certain clinical applications. Overcoming these impediments will be essential for stakeholders who want to take advantage of the considerable expansion prospects of the molecular diagnostics market.
How is Artificial Intelligence Transforming the Molecular Diagnostics Industry?
Artificial Intelligence (AI) and Internet of Things (IoT) are transforming the molecular diagnostics market by increasing speed, accuracy, and accessibility of disease diagnosis. AI can analyze large genomic datasets to detect anomalies and patterns, allowing for customized treatment regimens based on unique genetic profiles. At the same time, IoT technology allows data and monitoring in real time, with constant molecular data that, when analyzed by AI, produces more accurate diagnostics. This synergy not just speeds up diagnostic processes but also reaches high-tech healthcare solutions to remote and underserved areas.
One recent development illustrating this revolution is the innovation by Indian Institute of Technology, Indore (IIT-I) researchers who have created an innovative Quantum AI nanotechnology to detect early and accurately genetic mutations, such as those that cause cancer. This technology converges explainable AI with quantum transport, providing a low-cost, high-resolution solution for integrated DNA sequencing. It breaks the limitations of conventional sequencing technologies, which decode raw electric signals using sophisticated AI algorithms, allowing for precise identification of mutations and personalized treatment plans, thus revolutionizing the field of molecular diagnostics.
To get more insights on this market click here to Request a Free Sample Report
Global Molecular Diagnostics Market is segmented by Product, Test Type, Sample Type, Technology, Application, End User and region. Based on Product, the market is segmented into Reagents & Kits, Instruments and Services & Software. Based on Test Type, the market is segmented into Lab Tests and POC Tests. Based on Sample Type, the market is segmented into Blood, Serum, And Plasma, Urine and Other Sample Types. Based on Technology, the market is segmented into Polymerase Chain Reaction, Isothermal Nucleic Acid Amplification Technology, DNA Sequencing & Next-Generation Sequencing, In Situ Hybridization, DNA Microarrays and Other Technologies. Based on Application, the market is segmented into Infectious Disease Diagnostics, Oncology Testing, Genetic Testing and Other Applications. Based on End User, the market is segmented into Diagnostic Laboratories, Hospitals & Clinics and Other End Users. Based on region, the market is segmented into North America, Europe, Asia Pacific, Latin America and Middle East & Africa.
Among the pivotal segments, reagents & kits emerge as the largest contributor, wielding considerable influence over molecular diagnostics market trends and revenue streams. These fundamental factors contribute to the basic framework of a molecular diagnostic process and are indispensable in identifying and characterizing nucleic acids, proteins, and other molecular markers or biomarkers of different diseases. Due to rapidly increasing incidence of infectious diseases, genetic disorders, cancer around the world, the requirement of reagents and kits are high because of the need of right diagnosis in right time. Additionally, the progress in the biotechnology field and the shift from one-size-fits-all therapies to more individualized treatment promotes this segment, increasing the applicability of molecular diagnostic solutions in various types of healthcare services.
The molecular diagnostics market has experienced significant growth, especially in the instruments category, and is expected to be the fastest growing over the forecast period. These complex equipment and technology comprise a range of devices for conducting molecular testing such as PCR machines, DNA sequencers, and mass spectrometers pirit among others. In molecular diagnostics laboratories, instruments are the front-line in terms of acting as enablers, providing the appropriate tools and capabilities to healthcare professionals to selectively amplify and analyze and interpreted genetic material with incredible accuracy and speed.
The specific application of the molecular diagnostic instruments is further driven by the growing technological advancements and added commitments towards research which in turn have pressured the development of the molecular diagnostic instruments to achieve the higher throughput rates, automation as well as the multiplexing. In addition, point-of-care molecular diagnostic devices bring testing services to a wider population and decentralize testing further, which would help to increase the opportunities for the development of this segment in both developed and developing countries.
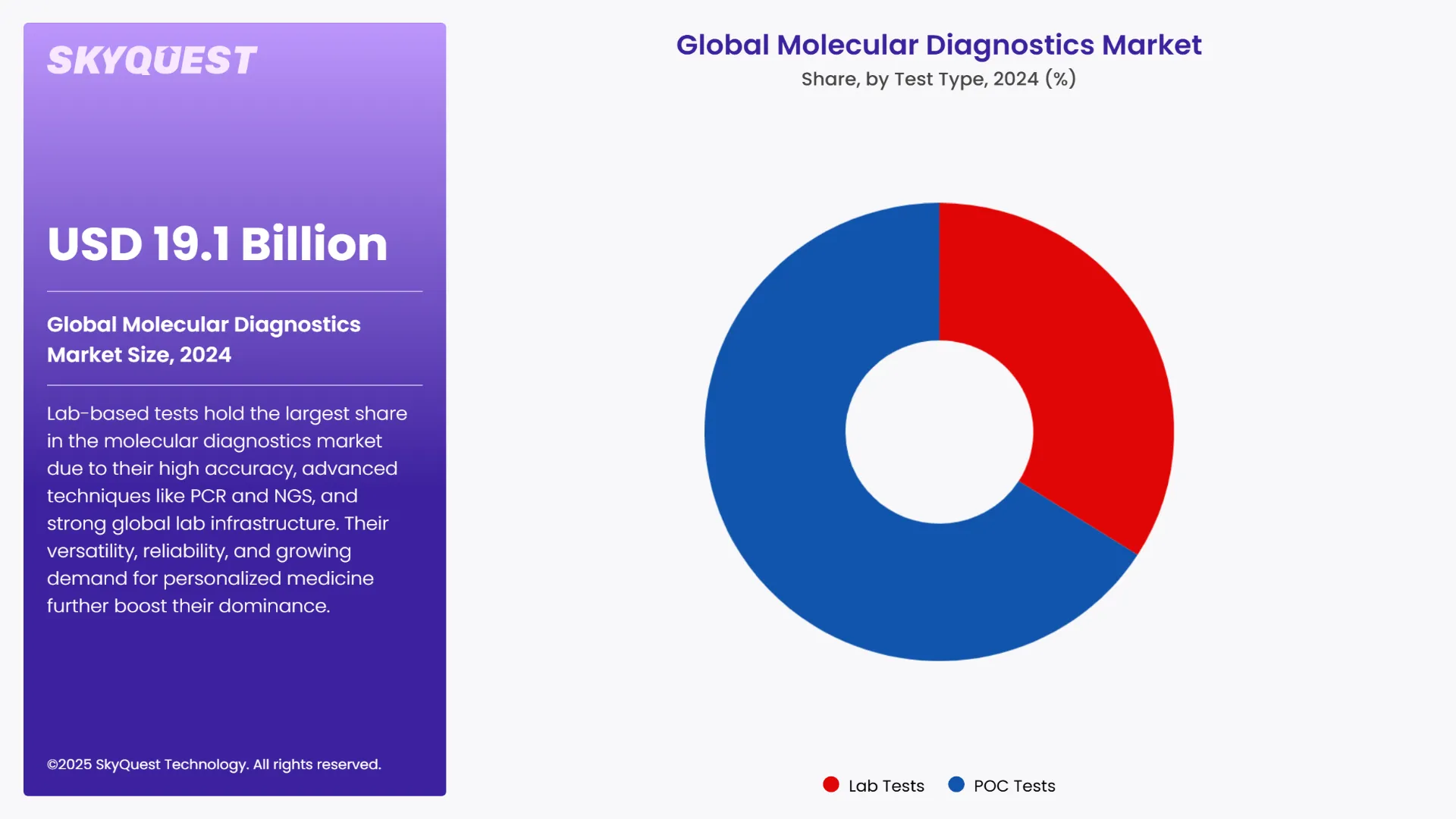
The lab tests take the largest molecular diagnostics market share in the global market. These tests are usually a more complicated procedure and are usually performed in specialized laboratories. This gives better results, and most of such tests are done in large central laboratories. Lab tests have many diagnostic features including PCR and next generation sequencing. This can be explained by a variety of conditions, which include comparatively high accuracy and repeatability of the examinations, versatility in terms of the tested materials, and well-developed framework for the large-scale laboratory centers all around the world. Furthermore, the shift in the molecular diagnostic industry toward Tiered pricing and Personalized Medicine as well as the growing need for better diagnostic platforms have added to the prominence of Lab-based Molecular Diagnostics segment that is currently considered the biggest segment of the molecular diagnostic market.
On the other hand, the fastest growing segment in the global molecular diagnostics market is PoC tests. This paper has depicted that there are certain advantages of PoC tests as compared to laboratory-based diagnosis methods and these are; fast diagnostic time, availability, and point of care screening. Such tests are aimed at providing notorious results promptly, sometimes at the exact location of the patient care, thereby being instrumental in timely decision making. Growth is driven by increasing demand for faster diagnostic tools especially in cases where a prompt response is necessary or in instances where tests can be done away from central laboratories such as with PoC molecular diagnostics.
Moreover, continuing improvements in miniaturization, automation of tests, and development of easy to use ‘point and click' platforms have enhanced continued widespread use of PoC testing devices in various regions of the globe and across multiple healthcare environments. Thus, it is the PoC segment in the market that becomes primarily associated with innovation and growth in the field of molecular diagnostics and determining the trends of shifts in the paradigms of diagnostic tests in the future.
To get detailed segments analysis, Request a Free Sample Report
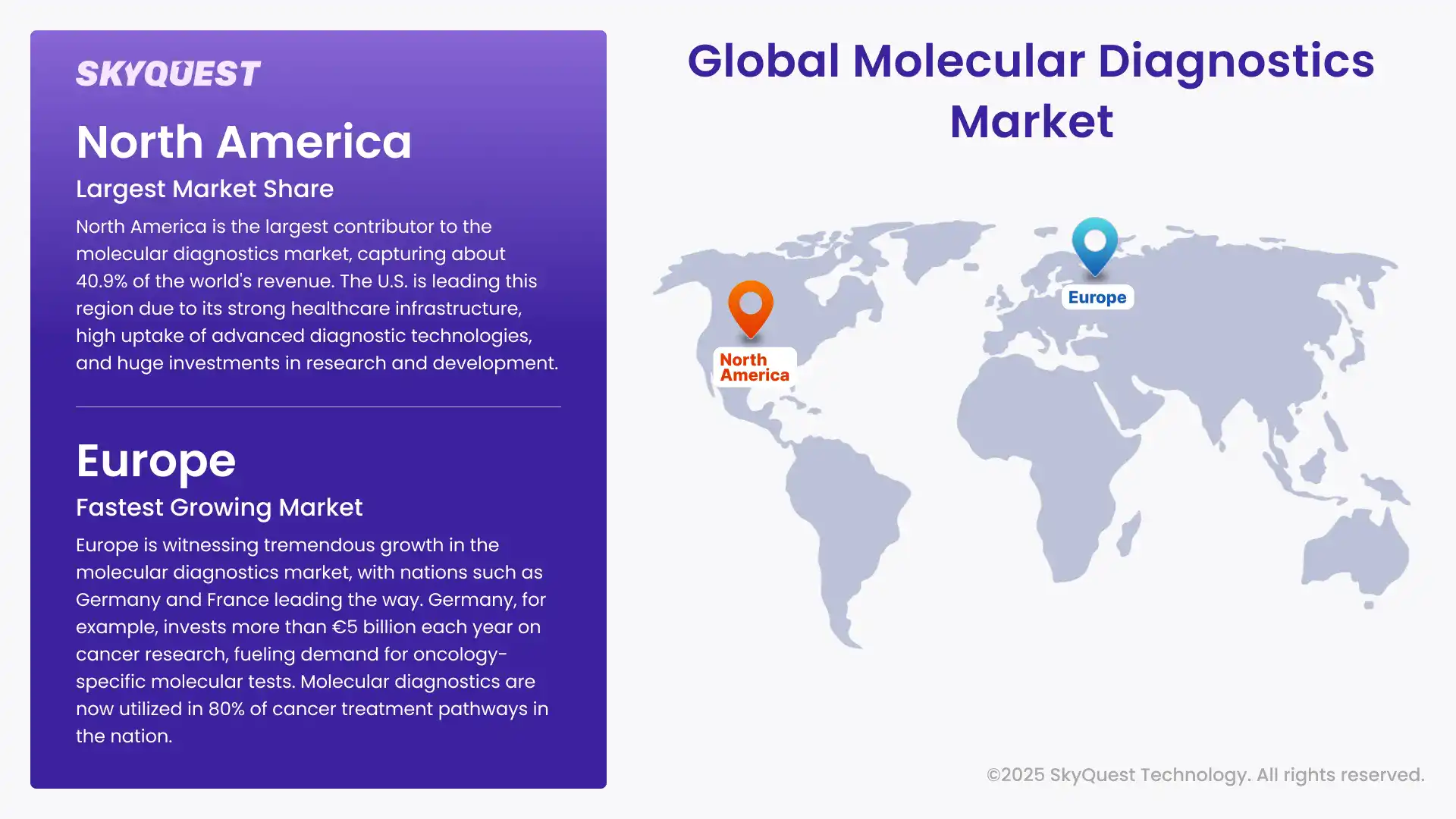
North America is the largest contributor to the molecular diagnostics market, capturing about 40.9% of the world's revenue. The U.S. is leading this region due to its strong healthcare infrastructure, high uptake of advanced diagnostic technologies, and huge investments in research and development.
United States dominates the molecular diagnostics market with around 87% of the local share. The country's dominance can be explained by a strong healthcare infrastructure, high level of adoption of cutting-edge diagnostic technologies, and presence of leading market participants. Recent news involves the FDA clearance of QIAGEN's NeuMoDx CT/NG Assay 2.0 in January 2024 to extend the company's test menu for its integrated PCR-based clinical molecular testing systems.
Canada is an emerging market in the molecular diagnostics space, which is expected to expand at around 5% CAGR during 2024-2029. The rising emphasis on preventive care and early detection of diseases by Canada's healthcare system has been one of the major drivers of growth. One of the recent developments includes the acquisition of Canada's LifeLabs by Quest Diagnostics at around C$1.35 billion to strengthen its foothold in the Canadian market.
Europe is witnessing tremendous growth in the molecular diagnostics market, with nations such as Germany and France leading the way. Germany, for example, invests more than €5 billion each year on cancer research, fueling demand for oncology-specific molecular tests. Molecular diagnostics are now utilized in 80% of cancer treatment pathways in the nation.
Germany dominates the European molecular diagnostics market through the likes of Siemens Healthineers and SYNLAB Group. Siemens Healthineers extended its oncology imaging capabilities with a €200 million buyout of Novartis's Advanced Accelerator Applications, adding to its PET radiopharmaceutical portfolio. SYNLAB, based in Munich, has more than 500 laboratories in Europe, conducting around 600 million tests in a year, and keeps expanding through selective acquisition.
France's molecular diagnostics sector is characterized by innovation in AI-based diagnostics. One French AI biotech firm, Owkin, has initiated a €33 million precision medicine initiative for cancer diagnosis and treatment using federated learning to train AI models without exposing proprietary data. This reduces data privacy risks while speeding diagnostic development.
The UK is a centre of genomic diagnosis and research, with Oxford Nanopore Technologies leading the way. The business works with the NHS to roll out fast pathogen screening at up to 30 locations, to be able to detect pathogens and pair treatments within six hours. Leica Biosystems also announced the move of its Companion Diagnostics and Advanced Assays division to Newcastle, further solidifying the UK's position in cancer diagnosis.
Asia Pacific region, the molecular diagnostics market is growing at a very fast pace, with India being anticipated to record the maximum CAGR between 2025 and 2030. The region had a revenue of USD 6,335.7 million in 2024 and is expected to reach USD 8,841.1 million by 2030 and grow at a CAGR of 5.5%. The growth is propelled by increased healthcare spending, rising awareness for disease detection at an early stage, and the incorporation of sophisticated diagnostic technologies.
China Dominating the market with a 22.8% share in 2022, China's lead is being driven by heavy investments in healthcare infrastructure and high-level molecular diagnostics capabilities. The health spending of the country grew to around US$1.1 trillion in 2023, much of which was invested in genetic testing centers and research. Leaders in the sector include players such as BGI and Hotgen Biotech, with Hotgen's self-test kits for COVID-19 antigen having EU CE clearance and FDA emergency use approval.
Japan has a high rate of diagnosis as well as good disease management practices. In 2023, the oncology-based molecular diagnostics market of Japan commanded a significant revenue share of 40.54%. Firms such as Sysmex and Fujirebio are developing local solutions, while Fujirebio is increasing its footprint through acquisitions and new assay launches for disease detection.
South Korea's molecular diagnostic market is supported by firms such as Seegene, which created and sold COVID-19 tests in the early days of the pandemic. Seegene's Allplex 2019-nCoV Assay was granted emergency use approval by the Ministry of Food and Drug Safety in February 2020, helping the nation respond to the pandemic effectively.
To know more about the market opportunities by region and country, click here to
Buy The Complete Report
Technological Advancements is Boosting the Molecular Diagnostics Market Growth
Increasing Prevalence of Infectious Diseases and Genetic Disorders
High Cost of Molecular Diagnostic Tests
Regulatory Challenges and Reimbursement Issues
Request Free Customization of this report to help us to meet your business objectives.
In the constantly shifting landscape of the molecular diagnostics market, companies are engaged in cut-throat competition and the process is characterized by innovations and strategies that can help the players gain a competitive edge over others. Some of the big players continue to dominate these markets through a broad portfolio of diagnostic services, mergers, and acquisitions, and broad sales and franchise networks. Also, the cooperation and collaboration between large-scaled companies and new entrant startups further intensify competition and innovation dynamics and the competition remained constant in bringing innovative technologies into the market thus maintaining a healthy market ecosystem with opportunities and threats simultaneously.
Shift towards Point-of-Care Testing
Integration of Artificial Intelligence (AI) and Big Data Analytics
SkyQuest’s ABIRAW (Advanced Business Intelligence, Research & Analysis Wing) is our Business Information Services team that Collects, Collates, Correlates, and Analyses the Data collected using Primary Exploratory Research backed by robust Secondary Desk research.
According to our analysis, global molecular diagnostics market is rising at a rapid rate and this is due to several factors these include technological developments, the rise in the incidences of diseases, and the rising trend in the consumer desire for customized treatment. Some of the factors that are driving growth in this market include the rising adoption of companion diagnostics, advancements in DNA sequencing technologies, and expanding applications in oncology, infectious diseases, and genetic testing. Furthermore, there is a rising regulated environment and decentralized testing, empowering point-of-care in the market and with the Patient Outcomes. Also, partnerships and alliances between major stakeholders, acquisitions, and strong emphasis on research and development are other factors that are fostering the market growth even further. Nevertheless, factors like a higher cost connected to molecular diagnostic procedures and a regulatory framework in emerging economies may act as a restraint in the growth of the market to a certain extent. In conclusion, due to the focus on diagnosing diseases at an early stage and targeted interventions (precision medicine), the global molecular diagnostics market is expected to continuously expand.
| Report Metric | Details |
|---|---|
| Market size value in 2024 | USD 19.1 Billion |
| Market size value in 2033 | USD 36.01 Billion |
| Growth Rate | 7.3% |
| Base year | 2024 |
| Forecast period | 2026-2033 |
| Forecast Unit (Value) | USD Billion |
| Segments covered |
|
| Regions covered | North America (US, Canada), Europe (Germany, France, United Kingdom, Italy, Spain, Rest of Europe), Asia Pacific (China, India, Japan, Rest of Asia-Pacific), Latin America (Brazil, Rest of Latin America), Middle East & Africa (South Africa, GCC Countries, Rest of MEA) |
| Companies covered |
|
| Customization scope | Free report customization with purchase. Customization includes:-
|
To get a free trial access to our platform which is a one stop solution for all your data requirements for quicker decision making. This platform allows you to compare markets, competitors who are prominent in the market, and mega trends that are influencing the dynamics in the market. Also, get access to detailed SkyQuest exclusive matrix.
Table Of Content
Executive Summary
Market overview
Parent Market Analysis
Market overview
Market size
KEY MARKET INSIGHTS
COVID IMPACT
MARKET DYNAMICS & OUTLOOK
Market Size by Region
KEY COMPANY PROFILES
Methodology
For the Molecular Diagnostics Market, our research methodology involved a mixture of primary and secondary data sources. Key steps involved in the research process are listed below:
1. Information Procurement: This stage involved the procurement of Market data or related information via primary and secondary sources. The various secondary sources used included various company websites, annual reports, trade databases, and paid databases such as Hoover's, Bloomberg Business, Factiva, and Avention. Our team did 45 primary interactions Globally which included several stakeholders such as manufacturers, customers, key opinion leaders, etc. Overall, information procurement was one of the most extensive stages in our research process.
2. Information Analysis: This step involved triangulation of data through bottom-up and top-down approaches to estimate and validate the total size and future estimate of the Molecular Diagnostics Market.
3. Report Formulation: The final step entailed the placement of data points in appropriate Market spaces in an attempt to deduce viable conclusions.
4. Validation & Publishing: Validation is the most important step in the process. Validation & re-validation via an intricately designed process helped us finalize data points to be used for final calculations. The final Market estimates and forecasts were then aligned and sent to our panel of industry experts for validation of data. Once the validation was done the report was sent to our Quality Assurance team to ensure adherence to style guides, consistency & design.
Analyst Support
Customization Options
With the given market data, our dedicated team of analysts can offer you the following customization options are available for the Molecular Diagnostics Market:
Product Analysis: Product matrix, which offers a detailed comparison of the product portfolio of companies.
Regional Analysis: Further analysis of the Molecular Diagnostics Market for additional countries.
Competitive Analysis: Detailed analysis and profiling of additional Market players & comparative analysis of competitive products.
Go to Market Strategy: Find the high-growth channels to invest your marketing efforts and increase your customer base.
Innovation Mapping: Identify racial solutions and innovation, connected to deep ecosystems of innovators, start-ups, academics, and strategic partners.
Category Intelligence: Customized intelligence that is relevant to their supply Markets will enable them to make smarter sourcing decisions and improve their category management.
Public Company Transcript Analysis: To improve the investment performance by generating new alpha and making better-informed decisions.
Social Media Listening: To analyze the conversations and trends happening not just around your brand, but around your industry as a whole, and use those insights to make better Marketing decisions.
REQUEST FOR SAMPLE
The global molecular diagnostics market is expected to arrive at a value of USD 33.7 billion by 2032, growing at a CAGR of 7.3% from 2025 and a growth from USD 19.1 billion in 2024.
Some of the key challenges include cost of testing and equipment, regulatory requirements, reimbursement, a shortage of skilled workforce, and consumer concerns about privacy and interpretation of complex genetic data.
Product segments include reagents & kits, instruments, and services & software, with reagents & kits leading the market and instruments growing the fastest due to the demand for high-throughput and automated diagnostics.
Technologies such as real-time PCR, next-generation sequencing (NGS), and DNA microarrays, have all changes the ways molecular diagnostics are used by enabling faster, more accurate, and sensitive detection of pathogens and genetic mutations.
Artificial intelligence can analyze large genomic datasets to identify aberrations. Other decisions surrounding treatment can be made in real-time with the aid of artificial intelligence combined with the Internet of Things (IoT). In this scenario patients can be monitored in real-time therefore improving access and clinical accuracy in even the most underserved communities.
Next-generation sequencing (NGS) is useful in molecular diagnostics as it offers high volume and comprehensive analysis of mutations which enables early diagnosis, personalized treatment and further advances and research tools particularly in oncology and infectious diseases.
In oncology molecular diagnostics is used to identify genetic mutations specific to cancers which allows detection of new cancers earlier, identify and guide therapies that will ultimately aid in diagnosing cancers and aid in personalizing treatment strategies for the cancer type identified.
It enables sensitive and accurate identification of infectious agents such as HIV, hepatitis, Mycobacterium tuberculosis (TB), that when identified accurately can enable faster treatment initiation and outbreak identification and which when used in precision medicine and assays such as polymerase chain reaction (PCR) or isothermal amplification improves infectious disease diagnosis and improve health outcomes in this population.
Personalized medicine is influencing demand for molecular diagnostics by requesting novel genetic also genomic information to customize therapies for individual patients. Personalized medicine is increasing the demand for and use of molecular diagnostic tests, particularly in oncology, infectious diseases, and inherited genetic conditions.
Traditional lab tests are more accurate. They are performed in centralized laboratories and involve a complex set of procedures, while point-of-care diagnostics can be performed quickly, and at the point of care for immediate feedback, which has driven demand and the growth of molecular diagnostics.
Want to customize this report? This report can be personalized according to your needs. Our analysts and industry experts will work directly with you to understand your requirements and provide you with customized data in a short amount of time. We offer $1000 worth of FREE customization at the time of purchase.
Feedback From Our Clients